
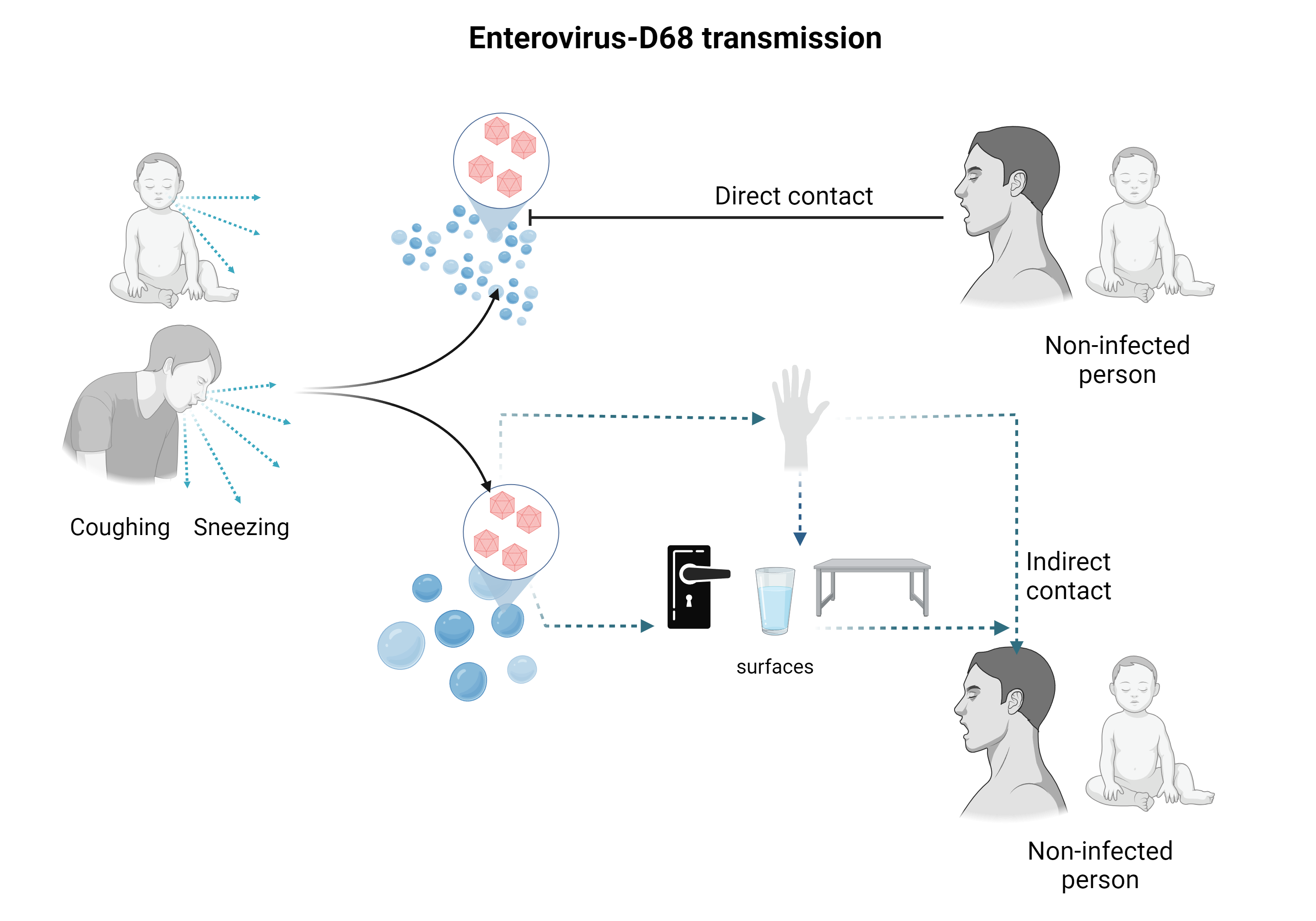
First described in 1962 in children hospitalized for pneumonia and bronchiolitis.
Since its discovery, during the long period of surveillance up to 2005, EV-D68 was reported only as a cause of sporadic outbreaks.
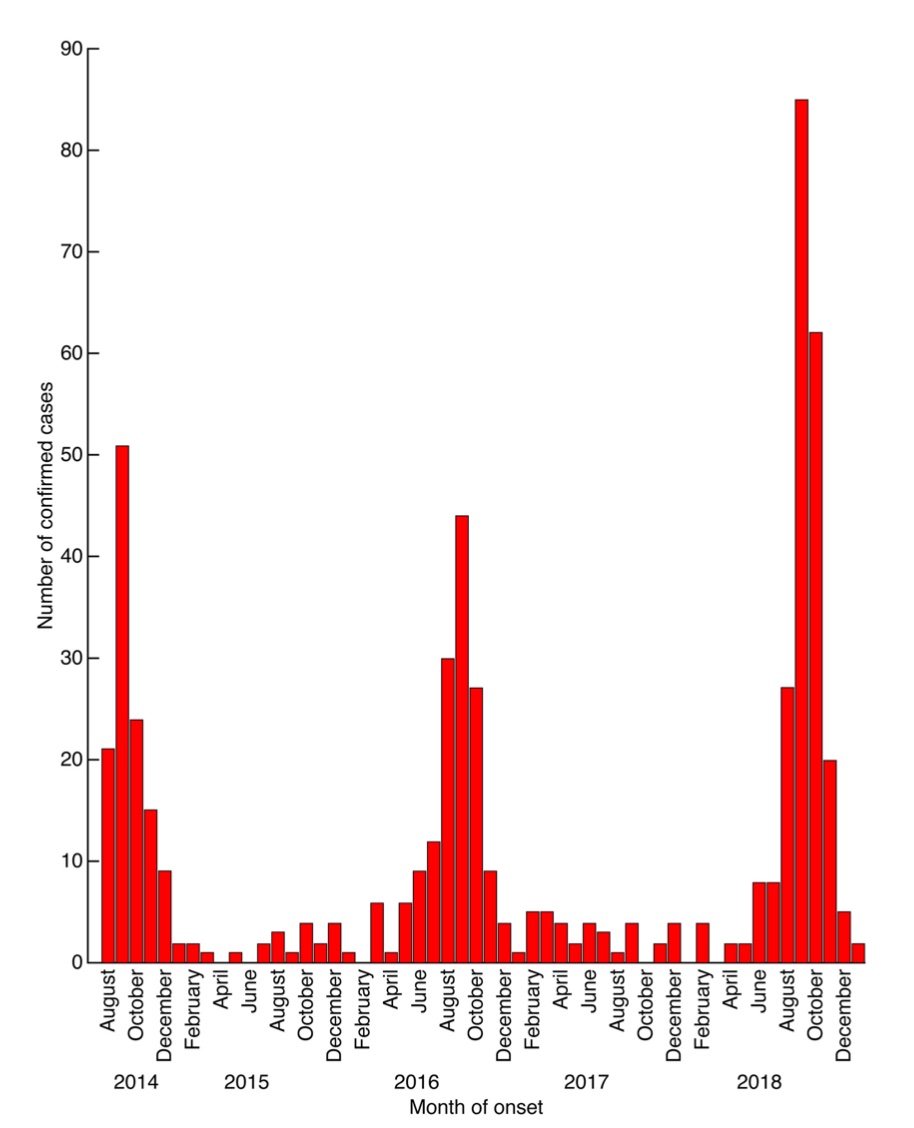
In August 2014, the United States and Canada experienced the first few cases of what resulted in the largest known EV-D68 outbreak in history. Unexpectedly, this outbreak of severe respiratory infections coincided with an upsurge in acute flaccid paralysis and cranial nerve dysfunction in children, which has caused concerns in the global community. Similar outbreaks were also seen in different European countries and Asian countries like Japan.
Subsequent EV-D68 epidemics were also seen in 2016 and 2018.
import { encodeTool } from '/_js/functions.js'
FA = FileAttachment
// Load the toolbox
families = await FA("/families.json").json()
_toolbox = await FileAttachment("/toolbox/toolbox.json").json()
toolbox = _toolbox.map( tool => encodeTool(tool) )
// console.log(toolbox)
addEncodedTools = (virus) => {
return ({ ... virus, tools: toolbox.flatMap( tool => virus[tool.encoded] ? tool.id : []) })
}
// Load all Newick tree file and create a dictionary `family -> tree`
nwks = await Promise.all(
families.map(async (family) => {
let tree = await FA("/"+ family + "/tree.newick").text()
return ({ family: family, nwk: tree })
}))
nwksAsMap = Object.fromEntries(nwks.map( el => [el.family, el.nwk] ))
// console.log(nwks)
// Load annotations for viruses for each family
_annotations = await Promise.all(
families.map( async (family) => {
let xls = await FA("/"+ family + "/family.xlsx").xlsx()
let _annotations = xls.sheet(0, { headers: true, range: ":L" })
let _trimmed = _annotations.map( virus => {
let trimmed = {}
Object.keys(virus).forEach( key => {
trimmed[key] = virus[key].trim()
})
return trimmed
})
return _trimmed.map( virus => ({... virus, family: family }) )
}))
// console.log(_annotations)
// Add tools information, just the list of tools ids
annotations = _annotations
.flat()
.map( virus => addEncodedTools(virus) )familyAnnotations = annotations
.filter(virus => virus.family == virusFamily)
function virusInfo(_family, _virus) {
const info =
familyAnnotations.filter(row => row.abbreviation == _virus && row.family == _family)
return info[0]
}
// Handle grouped viruses by extending the model with isGroup: false/true
addGrouped = (virus) => {
const isGroup = (typeof virus.group_abbreviation !== "undefined") && (virus.group_abbreviation !== "")
const virus_pointer = (isGroup) ? virus.group_abbreviation : virus.abbreviation
return (
{ ... virus,
isGroup: isGroup,
virusPointer: virus_pointer,
resVirusId: (isGroup) ? virus.group_abbreviation : virus.abbreviation,
resAbbreviation: (isGroup) ? virus.group_abbreviation : virus.abbreviation,
resVirusName: (isGroup) ? virus.group_virus_name : virus.virus_name
}
)
}
// Create a list of all viruses of interest in the Excel file
// Add group information early on
annotatedViruses =
familyAnnotations
.filter(virus => virus.virus_of_interest == "Yes")
.map(virus => addGrouped(virus))
// Create a sublist of viruses with toolbox
// Take into account groups by selecting unique entries
toolboxAnnotatedViruses =
annotatedViruses
.filter(virus => virus.availability_in_toolbox == "Yes")
.filter((value, index, self) => {
return self.findIndex(v => v.resAbbreviation === value.resAbbreviation) === index;
})
// Derive a virus object from the ID
// The ID can be either a virus_id (from the ictv tree) or a group_abbreviation
virusIdToVirus = (virus_id) => {
// lookup the virus_id in the full list of viruses
const fullList =
annotatedViruses
.filter(v => v.virus_id.replace(/^'+|'+$/g, '') == virus_id || v.abbreviation == virus_id)
//.filter(v => v.virus_id == virus_id || v.abbreviation == virus_id)
// lookup the abbreviation for the group
const groupList =
toolboxAnnotatedViruses
.filter(v => v.group_abbreviation == virus_id)
if (fullList.length > 0) {
return fullList[0]
}
if (groupList.length > 0) {
return groupList[0]
}
}
// Return all matches for a virus ID
virusIdToViruses = (virus_id) => {
// lookup the virus_id in the full list of viruses
const single_virus =
annotatedViruses
.filter(v => v.virus_id.replace(/^'+|'+$/g, '') == virus_id || v.abbreviation == virus_id)
[0]
if (single_virus.isGroup) {
return annotatedViruses
.filter(v => v.group_abbreviation == single_virus.group_abbreviation)
} else {
return [ single_virus ]
}
}
function renderVirusToolbox(family, virus_id) {
// First extract the virus object from the ID or abbreviation
// This takes into account the groups
const virus = virusIdToVirus(virus_id)
if (typeof virus.tools !== "undefined" && virus.tools.length > 0) {
return html`
${toolbox.filter(tool => virus.tools.includes(tool.id) ).map(tool =>
html`
<h2 id="${tool.id}-section">${tool.name}</h2>
<div class="container grid">
<div class="g-col-12 g-col-md-3 toolbox one-tool" style="text-align:center;">
<div id="toolbox-contents" class="tool-container">
<a href="/toolbox/index.html#${tool.id}-section">
<div class="tool-wrapper">
<div id="${tool.id}" class="tool">
<div class="tool-tooltip-text">${tool.name}</div>
${tool.icon.map( i =>
html`<img class="tool-icon" height="${(tool.icon.length > 1) ? 150/tool.icon.length : 100}%" src="${i}"/>`
)}
</div>
</div>
</a>
</div>
</div>
<div class="g-col-12 g-col-md-9">
<p>${tool.description}</p>
</div>
<div class="g-col-1" style="text-align:center;">
</div>
</div>
<div class="col-xs-12" style="height:20px;"></div>
`
)}
`
}
else {
return html`
<p> </p>
`
}
}